In the world of geology, no rock type records our planet’s biological and chemical history quite like limestone. While it forms the foundation of our daily lives—from the construction industry to cement manufacturing—it holds a much deeper significance for exploration geologists: It is a massive ore trap.
In this guide, we will explore what limestone is, the environments where it forms, its main varieties, and why it hosts world-class mineral deposits (such as Skarns, MVT, and Carlin-type).
Part 1: What is Limestone?
Limestone is a chemical or biochemical sedimentary rock composed primarily of calcium carbonate (CaCO3) in the form of calcite and aragonite minerals. Unlike most clastic sedimentary rocks (like sandstone or shale), limestone is typically not formed from detrital material transported from land, but rather from materials produced within the water bodies themselves (especially oceans).
The most classic field method for identifying limestone is the “acid test.” When a drop of dilute hydrochloric acid (HCl) is applied, the calcite mineral reacts, fizzing and effervescing vigorously due to the release of CO2 gas.
Part 2: Environments and Formation of Limestone
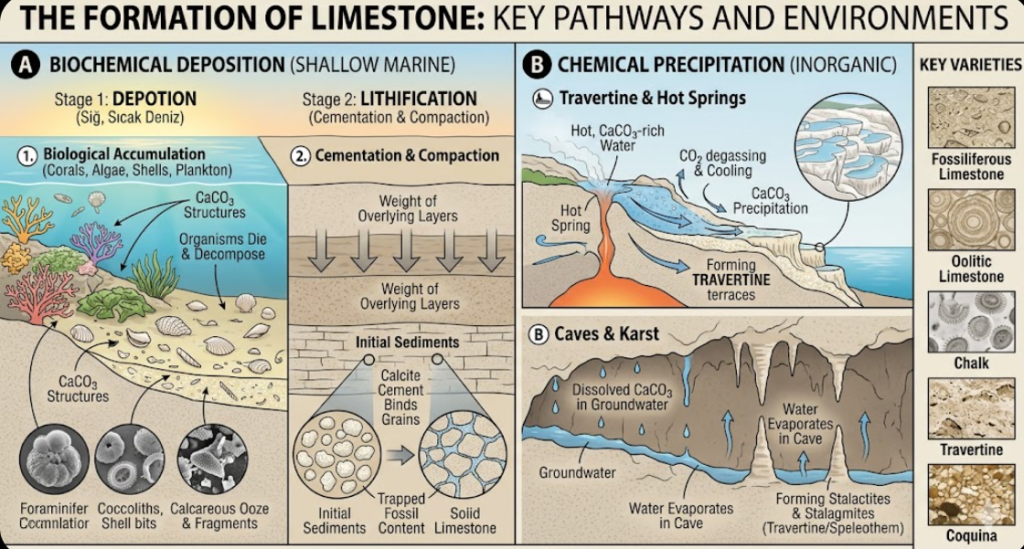
Specific environmental conditions, often referred to as “Carbonate Factories,” are required for limestone to form. These rocks develop through two main mechanisms:
A. Biochemical (Organic) Environments
The vast majority of the world’s limestones are the product of biological activity.
- Shallow, Warm Tropical Seas: This is the ideal environment for limestone formation (similar to the modern Bahamas or the Great Barrier Reef). In clear, warm, and shallow waters reached by sunlight, corals, algae, bivalves, and foraminifera produce calcium carbonate shells. When these organisms die, their shells accumulate on the seafloor, compacting over time to form fossiliferous limestones.
- Deep Marine (Pelagic) Environments: Formed by the continuous “snowfall” of the calcareous shells of microscopic plankton (such as coccolithophores) onto the ocean floor.
B. Chemical (Inorganic) Environments
Formed without biological activity, directly through the supersaturation and precipitation of calcium carbonate from water.
- Caves and Karst Environments: Forms stalactites, stalagmites, and dripstones through the evaporation of groundwater and pressure changes.
- Thermal Springs: As hot, mineral-rich waters reach the surface at hot springs, they rapidly cool and lose gas, causing calcium carbonate to precipitate (the travertine terraces of Pamukkale in Turkey are a classic example).
- Evaporitic Basins: Precipitates chemically through the evaporation of shallow lakes in arid climates.
Part 3: Types of Limestone

Limestones are classified into several subtypes based on their depositional environment and components:
- Fossiliferous Limestone: Contains abundant, visible macrofossils (such as corals, brachiopods, and crinoids).
- Oolitic Limestone: Composed of small, spherical grains (oolites) formed in shallow, high-energy wave environments by the concentric accumulation of calcite layers (like an onion) around a sand grain or shell fragment.
- Micrite (Carbonate Mud): Extremely fine-grained limestone formed in very calm, low-energy environments. The grains are too small to be seen with the naked eye.
- Chalk: A very soft, porous, and white limestone made entirely of the microscopic shells of marine organisms (coccoliths).
- Coquina: A highly porous limestone composed almost entirely of broken, fragmented, and poorly cemented seashells. It indicates a high-energy beach environment.
- Travertine: A banded limestone formed by inorganic precipitation at freshwater springs, particularly hot springs.
Part 4: Importance for Mineralization (Why Are They Economic Targets?)
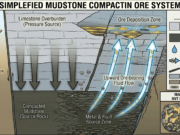
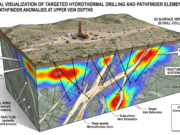
An exploration geologist gets highly excited when encountering a massive limestone formation in the field (especially if it is faulted or intruded by a magmatic body). This is because limestone is one of nature’s most perfect ore traps. There are three main physical and chemical reasons for this:
1. Massive Chemical Reactivity (The Sponge Effect)
Limestone (CaCO3) has a basic character. Most hydrothermal fluids (ore-bearing waters) rising from the depths are acidic. The moment an acidic, metal-laden hydrothermal fluid comes into contact with limestone, a violent chemical reaction begins. The limestone neutralizes the acid, causing the fluid to instantly drop its carried metals (Gold, Silver, Lead, Zinc, etc.) out of solution, precipitating them directly into the limestone to form rich deposits.
2. The Engine of Skarn Deposits
When a hot magma body (like a granite pluton) intrudes and cuts through underground limestone blocks, a massive transfer of heat and fluids occurs. As the pure limestone “bakes” and metamorphoses into marble, it reacts with the silica and metals introduced from the magma, forming Skarn Deposits. Many of the world’s largest copper, gold, tungsten, and iron deposits are found within these calc-silicate alteration zones (limestone contacts).
3. Porosity and Karstification (Reservoir Rock Properties)
Limestone is easily dissolved by groundwater (the karstification process). Over time, massive caves, voids, and underground river networks form within the limestone. These open spaces (breccia zones) create perfect storage areas for ore-bearing fluids that enter the system later.
- MVT (Mississippi Valley Type) Deposits: Some of the world’s most important sources of Lead and Zinc, MVT deposits precipitate within these dissolution cavities in limestones and dolomites.
- Carlin-Type Gold Deposits: Particularly notable in Nevada, hydrothermal fluids dissolve and remove the carbonate rock (limestone) at a microscopic level, replacing it with a spongy texture hosting microscopic “invisible gold.”
In summary; Limestone is not just a graveyard of ancient seas, but thanks to its reactive nature and solubility, it is one of the richest chemical laboratories in the Earth’s crust.